50%
Improvement in Overall Operational Efficiency
60%
Reduction in Repetitive Workload
$450,000+
Saved in Direct EDC Cost Savings
As one of the top 50 global pharma giants, CSPC Pharmaceuticals is on a mission to innovate and deliver life-saving drugs. With over $500 million invested in R&D annually, CSPC is leading the charge in clinical trials. Their vision is clear: to create better medicine and contribute to the health of mankind.

At the heart of this mission is a massive investment in research and development. CSPC invests over $500 million annually in R&D, with a team of over 2,000 researchers working on more than 300 clinical trial projects. Clinical trials are the lifeblood of the pharmaceutical industry, the long and arduous process of testing new drugs for safety and efficacy. They are also incredibly expensive, time-consuming, and complex.
For years, CSPC, like many of its peers, managed this critical process using a patchwork of traditional, manual methods. But in an era of fierce competition and mounting pressure to control costs, the company knew that this old way of working was no longer sustainable. They were facing a critical bottleneck that threatened to slow down their innovation engine and put them at a competitive disadvantage.
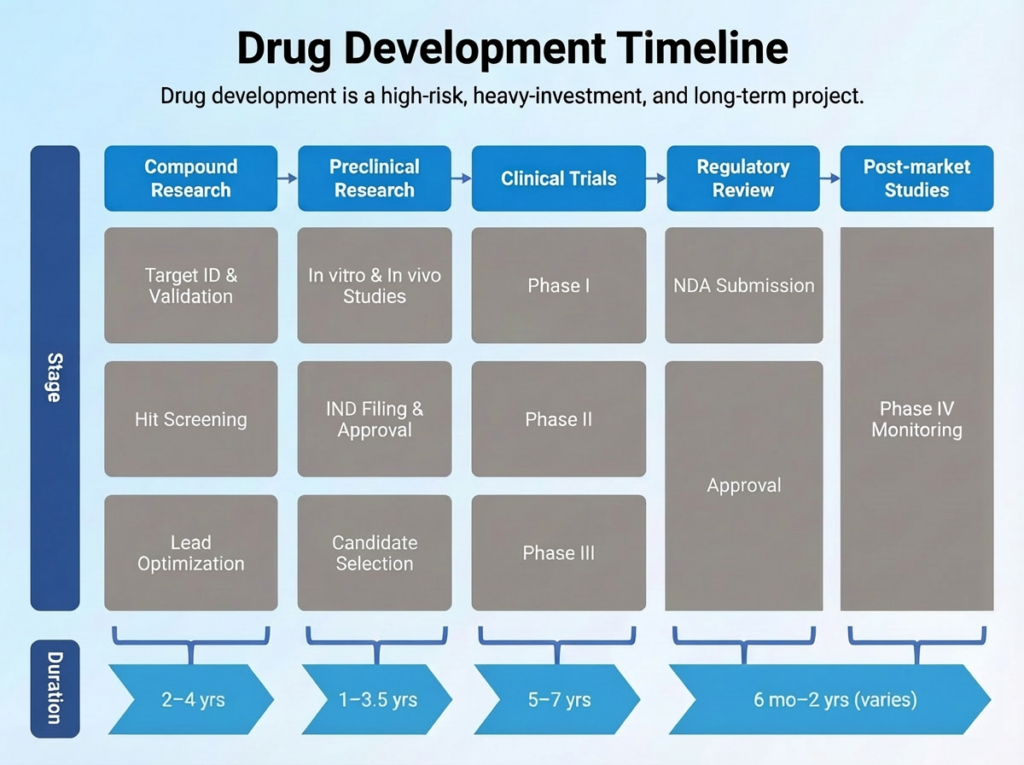
This is the story of how CSPC’s clinical trial team took matters into their own hands. They didn’t wait for a massive, top-down IT overhaul. Instead, they adopted a nimble, three-step strategy, using a no-code platform to build a powerful digital engine for their clinical trial operations. They have not only streamlined their processes and slashed costs, but they have also created a new model for digital innovation that is setting a new standard for the entire industry.
The Challenge: Three Bottlenecks in the Race for a Cure
CSPC’s leadership team identified three critical bottlenecks that were slowing down their clinical trial process, driving up costs, and putting their R&D pipeline at risk.
The Data Disconnect: Drowning in a Sea of Spreadsheets
Clinical trials are a team sport, involving dozens of partners, from research centers and labs to patients and regulatory agencies. At CSPC, the data from all these different sources was scattered across a multitude of disconnected systems—EDC (Electronic Data Capture), CTMS (Clinical Trial Management System), and many others. This created a massive data disconnect.
To get a complete picture of a trial’s progress, the clinical research associates (CRAs)—the frontline workers who manage the trials—had to manually pull data from all these different systems and consolidate it into Excel spreadsheets. The process was a nightmare. Different departments wanted the data sliced in different ways, so the CRAs were constantly re-entering the same information into multiple spreadsheets. The work was tedious, repetitive, and prone to errors. It was a classic case of “Excel hell,” and it was burning out the company’s most valuable employees.
The Information Lag: Making High-Stakes Decisions with Outdated Data
The reliance on manual, spreadsheet-based reporting created a dangerous information lag. By the time the data was collected, consolidated, and presented to management, it was often a week out of date. In the fast-moving world of clinical trials, a week is an eternity. A problem with patient recruitment or a quality issue at a research site could fester for days before anyone in a position to do something about it even knew it was happening. By the time a decision was made, it was often too late to fix the problem effectively.

This lack of real-time data made it impossible for leaders to get a clear, up-to-the-minute view of their portfolio. They were flying blind, making multi-million-dollar decisions based on a fuzzy, outdated picture of reality. It was a risky way to run a business, and it was costing the company dearly in wasted resources and missed opportunities.
The Cost Control Puzzle: A Black Hole of R&D Spending
Drug development is one of the most expensive endeavors on the planet, and controlling costs is a constant battle. At CSPC, the fragmented data and manual processes made it nearly impossible to get a clear handle on the true costs of their clinical trials. It was difficult to track the spending on materials, manpower, and other resources for each project, and even harder to compare the actual spending against the budget.

This lack of financial visibility was a major problem. It made it difficult to assess the return on investment for each trial, to identify areas of waste, and to make smart decisions about where to allocate resources. The R&D budget was a black hole, and the company knew that it was leaving millions of dollars on the table.
The Solution: A Three-Step Strategy for Digital-Driven Drug Development
Faced with these challenges, CSPC’s clinical trial team didn’t wait for a top-down mandate or a massive IT project. They took an agile, business-led approach, creating a three-step strategy to build their own digital solutions from the ground up. Using Jodoo as their core platform, they began to dismantle their biggest bottlenecks systematically.
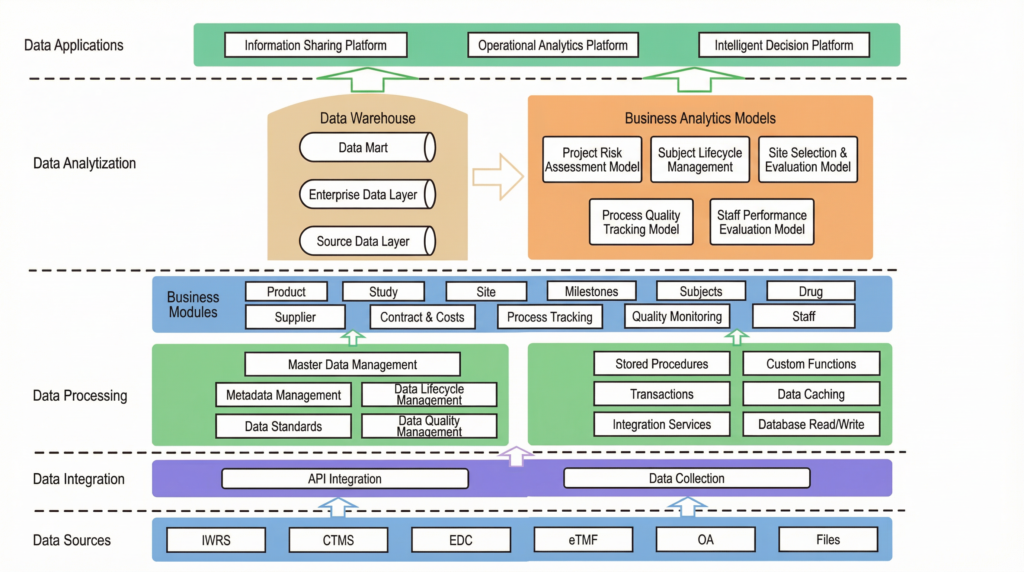
Step 1: Build an Information Sharing Platform (The Single Source of Truth)
The first step was to solve the data disconnect. The team designed and built a central data warehouse to act as a single source of truth for all clinical trial data. Using Jodoo’s flexible integration capabilities, they built bridges to all their existing systems—EDC, CTMS, and others—and created automated workflows to pull all the data into one place.
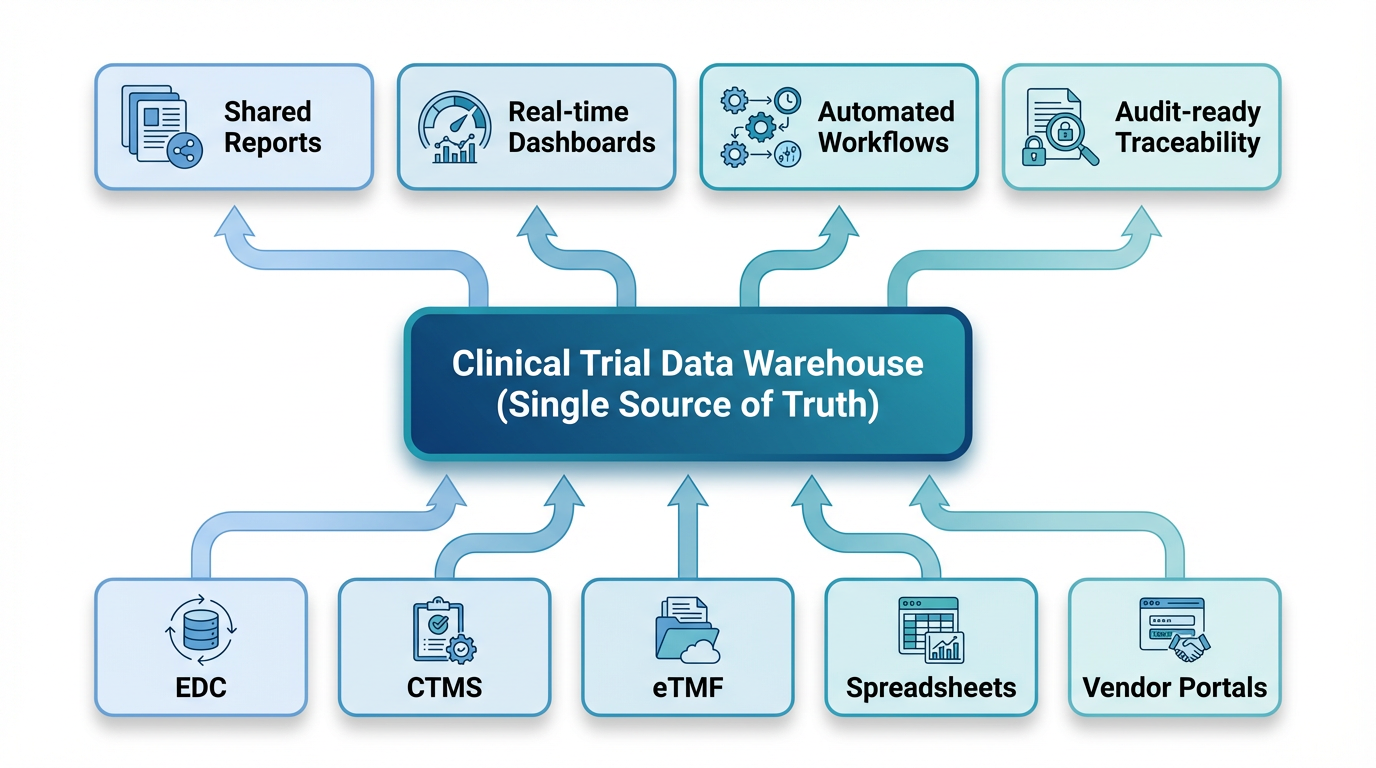
For any data that couldn’t be pulled automatically, they built simple, user-friendly Jodoo apps to capture it. This completely eliminated the need for manual data entry in spreadsheets. Now, data only needs to be entered once, and it is instantly available to everyone who needs it, in a standardized, reliable format. The “Excel hell” was finally over.
Step 2: Build an Operations Analysis Platform (From Data to Decisions)
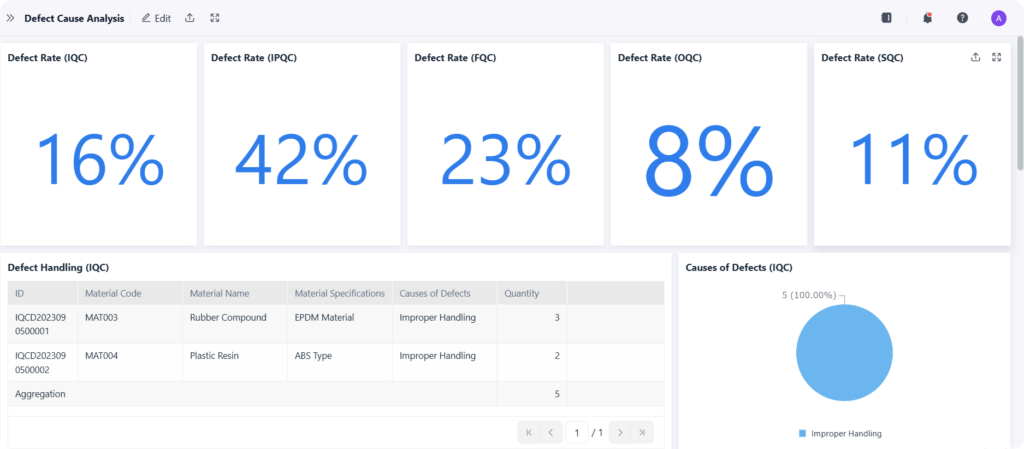
With all their data in one place, the next step was to make it useful. The team used business intelligence (BI) tools to build a suite of powerful, interactive dashboards on top of their new data warehouse. This was the Operations Analysis Platform, and it transformed the way the company made decisions.
They created different dashboards for every level of the organization:
- For Executives: A high-level “cockpit” view that shows the overall health of the entire clinical trial portfolio—tracking progress, budget, and key milestones at a glance.
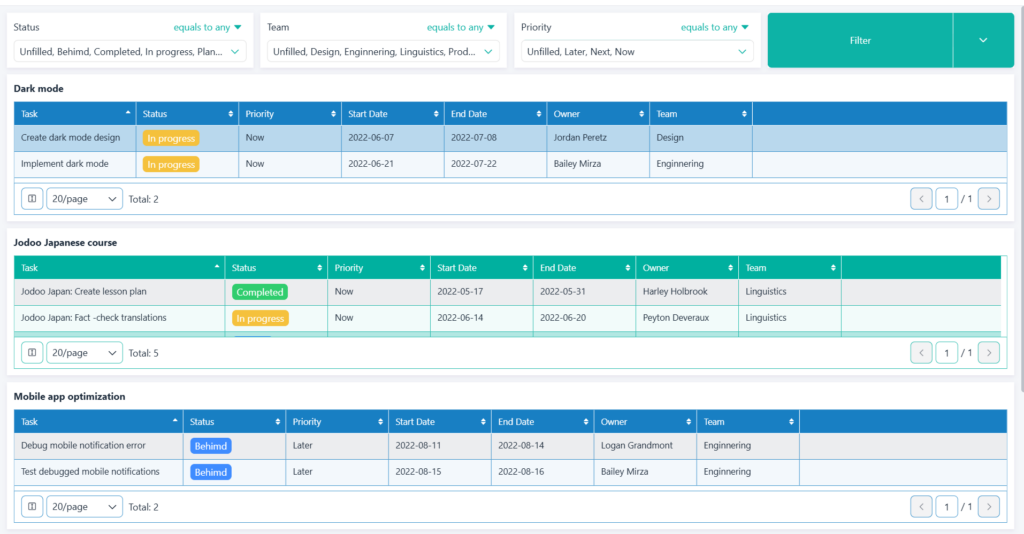
- For Managers: Detailed dashboards that allow them to drill down into specific trials, analyze performance by region or research center, and identify potential problems before they escalate.
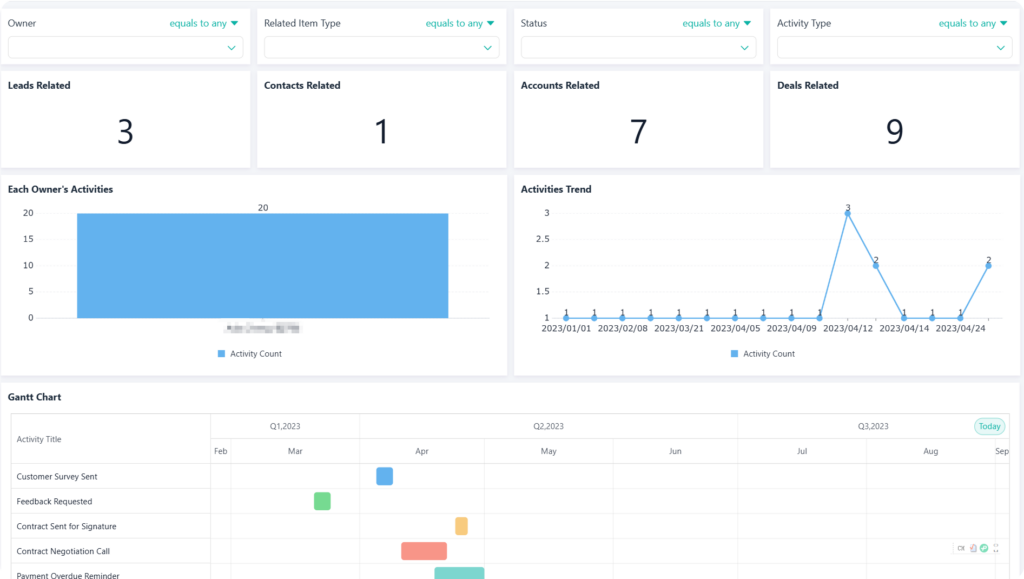
- For Frontline Workers: Self-service analytics tools that empower CRAs to explore the data themselves, helping them manage their own work more effectively and contribute to improving the process.
This platform replaced the slow, static, weekly PowerPoint reports with live, real-time data. Everyone, from the CEO to the CRA, was finally looking at the same numbers, at the same time.
Step 3: Build A “Lightweight” EDC System
One of the most powerful examples of this new approach was the creation of a “lightweight” Electronic Data Capture (EDC) system using Jodoo. Full-featured EDC systems from global vendors are incredibly powerful, but they are also extremely expensive—costing millions of dollars—and can be slow to set up.
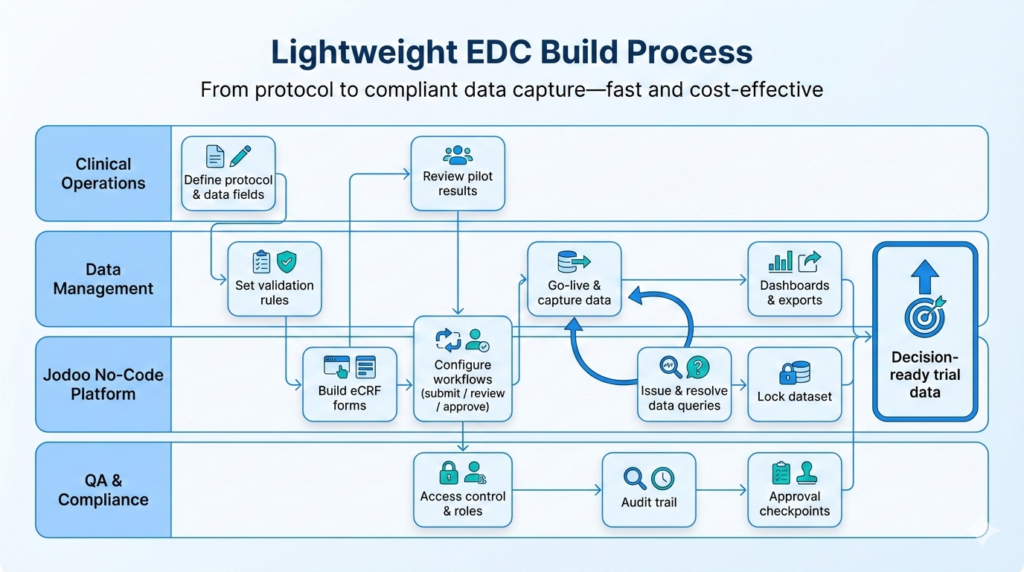
The CSPC team realized that for many of their simpler, less complex trials, they didn’t need all the bells and whistles of a full-scale EDC. So, they used Jodoo to build their own. They created a standardized, templatized version that covered 85% of the functionality they needed. Now, instead of spending months and a huge amount of money to set up a new EDC for a simple trial, they can simply copy their Jodoo template and have a new system up and running in a fraction of the time.
This single innovation has been a game-changer. It has dramatically accelerated the startup time for new trials and, as a supplement to their existing EDC systems, has saved the company over $450,000 in licensing fees alone.
The Results: A Healthier Bottom Line and a Faster Path to Market
The impact of CSPC’s three-step digital strategy has been nothing short of transformative. By empowering their business teams to solve their own problems, the company has not only streamlined its clinical trial operations but has also unlocked significant financial savings and accelerated its timeline for bringing new drugs to market. The results are a powerful demonstration of what is possible when you put the right tools in the hands of the right people.
| Key Performance Indicator | Before Jodoo | After Jodoo |
|---|---|---|
| Overall Operational Efficiency | Slow, manual, and reactive | 50% improvement |
| Repetitive Workload | High, causing employee burnout | 60% reduction |
| Manual Data Entry | 4x the workload | Reduced to 1/4 of previous levels |
| Data Utilization Efficiency | Low, data trapped in silos | 80% improvement |
| EDC Setup Time (Simple Trials) | Weeks or months | Reduced to 1/5 of the previous time |
| Direct EDC Cost Savings | N/A | Over $450,000 saved |
| Financial Risks Averted | Difficult to quantify | $1.5 Million |
The numbers tell a compelling story. A 50% improvement in overall operational efficiency means that trials are running faster and more smoothly than ever before. The 60% reduction in repetitive work and the dramatic cut in manual data entry have freed up the company’s highly skilled CRAs to focus on what they do best: managing the quality and integrity of the trials. This has led to an 80% improvement in data utilization efficiency, as data is no longer just being collected for regulatory purposes; it’s being used to drive better, faster decisions.
The creation of the lightweight EDC system has been a huge financial win, saving the company over $450,000 in direct costs. But the financial impact goes even further. By providing real-time visibility into the financial health of each trial, the new platform has helped the company identify and mitigate an estimated $1.5 million in financial risks, such as budget overruns and inefficient resource allocation.
Perhaps the most important results are the ones that are hardest to quantify. By improving the efficiency of center screening and patient recruitment, the company has been able to increase patient enrollment rates by nearly 20% and reduce patient dropout rates by 5%. In the world of clinical trials, these numbers are huge. They mean that trials can be completed faster, and that new, life-saving drugs can get to the patients who need them sooner.
Future Outlook: Building an Intelligent Decision-Making Platform
For CSPC, this is just the beginning. The success of their three-step strategy has laid the foundation for their ultimate vision: to build a fully integrated, intelligent decision-making platform for their entire clinical trial portfolio. This is Step 3 of their strategy, and it is already in the works.
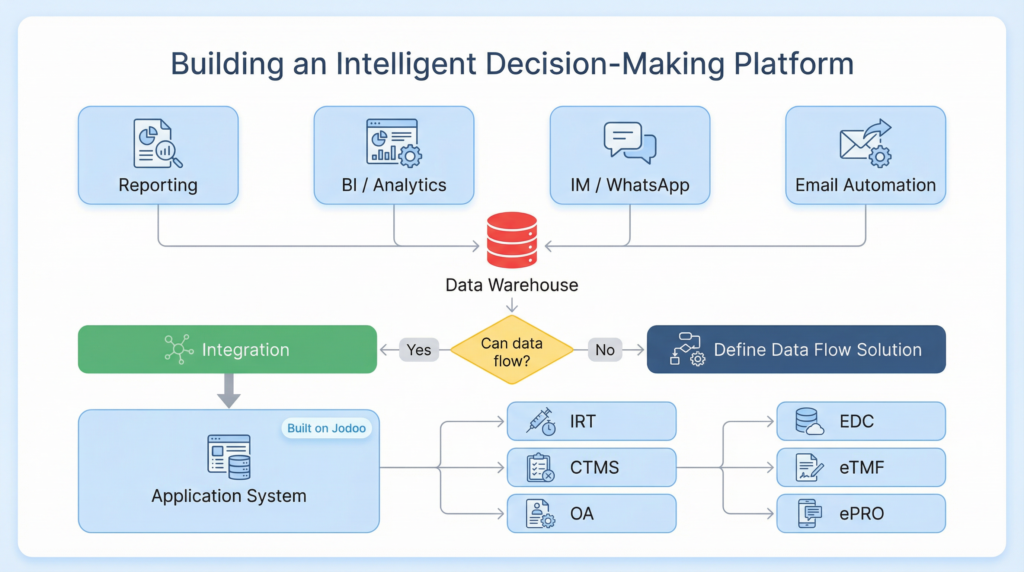
The goal is to move from simply analyzing what has happened in the past to predicting what will happen in the future. The team is developing sophisticated algorithms that will run on top of their data warehouse, automatically identifying potential risks and opportunities. For example, the system will be able to predict which research centers are likely to be the most successful at recruiting patients for a particular type of trial, or which projects are at risk of going over budget.
These insights will be automatically pushed out to the relevant team members, triggering a proactive response. This will create a continuous feedback loop—from data to insight to action to new data—that will allow the company to constantly learn and improve. As the project leader explained, “We are not professional IT developers; we are data analysts who are focused on the business. Our goal is to use these powerful tools to solve real business problems and create a platform that will allow us to make smarter, faster decisions.”
CSPC’s story is a powerful example of how a large, established company can drive digital innovation from the bottom up. They have shown that you don’t need a massive, multi-year IT project to achieve transformative results. By embracing a culture of agile, business-led development, they have not only improved their own performance but have also created a new blueprint for the future of drug development. They are not just making good medicine; they are building a better, faster, and more efficient way to bring that medicine to the world.
Ready to start your own digital transformation journey?



